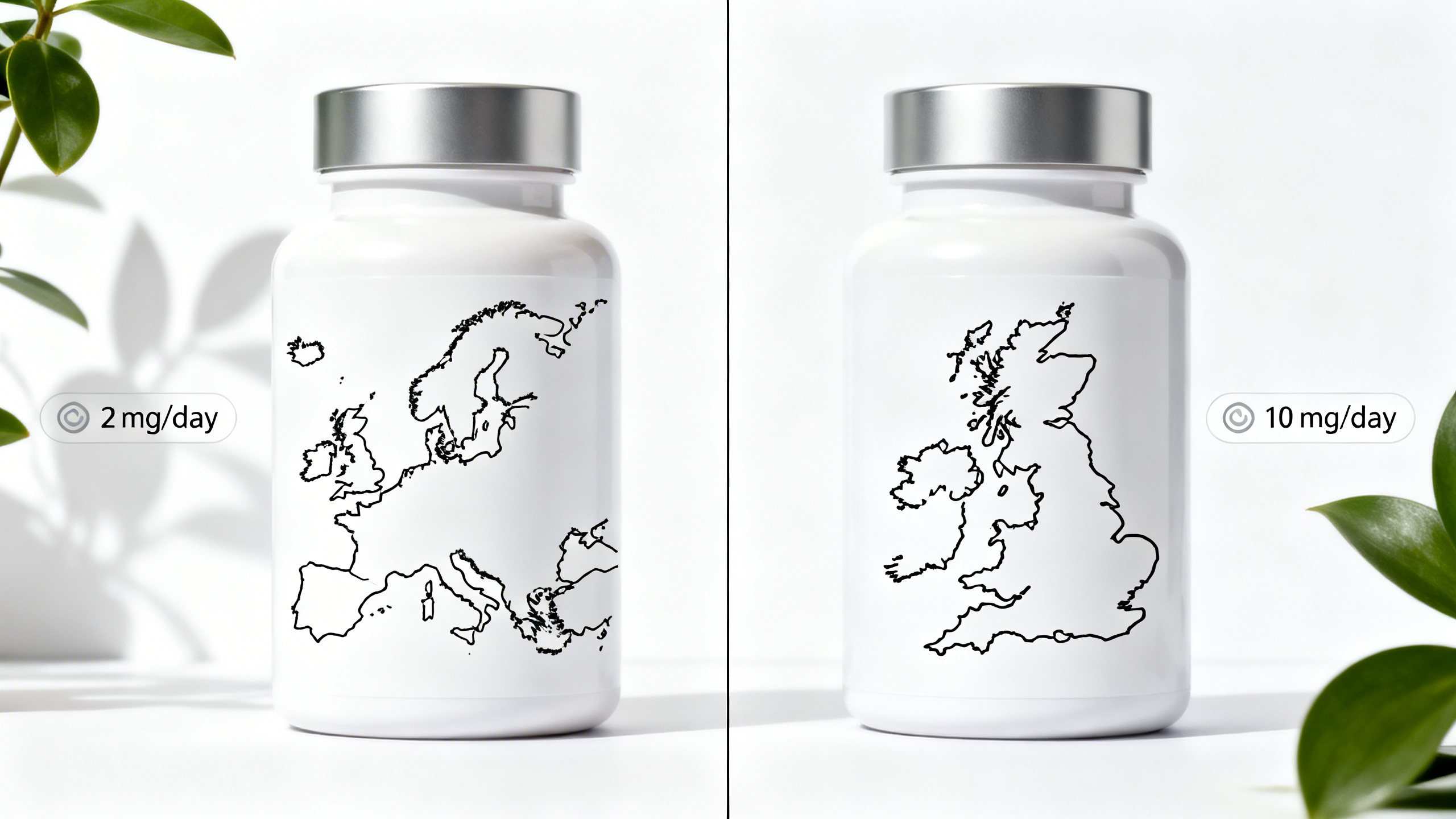
E‑commerce has made it easy to sell the same ingestible CBD product into multiple jurisdictions—but food law does not travel as easily as parcels do. As of early 2026, operators targeting both the European Union and Great Britain face a practical contradiction in intake messaging:
For cross‑border brands, this is not just an academic debate. It affects label serving sizes, directions for use, pack architecture, online product pages, retailer onboarding, and—critically—how you defend your products in audits or enforcement actions.
This article is informational only and not legal advice.
In its updated work on CBD safety as a novel food, EFSA’s NDA Panel set a provisional safe intake level of 0.0275 mg/kg bw/day (approximately 2 mg/day for a 70 kg adult), explicitly noting that data gaps remain and that this provisional level may be revisited when better toxicology and human data become available.
EFSA’s position matters because EFSA is the EU’s risk assessor for novel foods under Regulation (EU) 2015/2283, and its scientific outputs heavily influence:
Key source:
The UK FSA (England/Wales) continues to encourage businesses to label products to reflect current consumer advice, including a 10 mg/day CBD intake for healthy adults.
The FSA’s current approach is embedded in a broader UK compliance framework that includes:
Key sources:
It is tempting to describe this as “EU stricter, UK looser.” In reality, the split reflects different regulatory moments:
The result: one consumer marketplace, two intake narratives.
Under EU novel foods rules, a product that meets the novel food definition cannot be legally marketed without authorization and inclusion on the Union list of authorized novel foods (Commission Implementing Regulation (EU) 2017/2470). The Commission’s authorization process and the Union list are here:
Even without EU‑wide authorizations, enforcement happens through national competent authorities applying:
Sources:
A key practical indicator is the EU’s Rapid Alert System for Food and Feed (RASFF), where notifications continue to flag ingestible CBD as an unauthorized novel food in specific products.
Example notifications:
If you are marketing a product as a food supplement, you still need to align with:
Practical EU supplement label expectations include a recommended daily portion and a warning not to exceed the stated portion (Directive 2002/46/EC).
Consumer-facing summary (EU):
Commission page on nutrition/health claims framework:
For England and Wales, the FSA’s Register of CBD products linked to novel food applications (often called the Public List) is the reference point many retailers use to decide what stays on shelves.
The FSA also explicitly states that enforcement decisions remain with local authorities—but the agency has recommended that products with certain statuses may stay on the market pending further consideration.
In August 2025, the FSA opened a public consultation on the authorisation of three CBD food products as novel foods, with the consultation closing 20 November 2025.
Even if you are not one of the applicants, this matters because it signals how UK risk managers may frame:
Do not overlook marketing compliance. UK regulators draw a hard line between foods/supplements and medicines. If you imply treatment or prevention of disease, you risk being treated as a medicine.
The operational problem is simple: your label’s “daily intake” language can be used against you.
If you run one pan‑regional SKU with a single set of directions, you risk:
The safest path for e‑commerce is to keep formulation constant (where possible) but tailor:
This can be executed via:
Important: within the EU, you must be cautious with geo‑targeting practices. The EU Geo‑blocking Regulation restricts unjustified discrimination, but it does not force you to sell every product into every country. You can still define delivery destinations and manage compliance risk.
Background:
At minimum, most cross‑border operators now need two intake tracks:
Operationally, this means:
The biggest hidden compliance issue is not the mg/day text—it’s that your unit dose can make compliance impossible.
If your single gummy or capsule contains 10–25 mg, you cannot credibly present an EU‑aligned serving suggestion near 2 mg/day without creating awkward fractional directions (“take 1/5 of a gummy”). That invites consumer misuse.
Common mitigation tactics:
Across both EU and UK, sophisticated retailers are increasingly asking for a “compliance dossier” that connects:
EFSA’s updated position emphasizes continuing uncertainty and the need to revisit conclusions when new data emerges. You should expect more questions about:
Reference EFSA output:
A frequent enforcement trigger is not the mg/day statement—it’s marketing copy that implies therapeutic benefit.
Key references:
Even when your focus is intake limits, enforcement often pivots to contaminants and classification.
For example, EU rules set maximum levels for certain contaminants in food, including specific contexts for hemp seed products under Commission Regulation (EU) 2023/915.
Member States may also have overlays that are stricter than EU baselines for certain product types.
EFSA has been clear that the level is provisional and dependent on future data. For operators, the actionable point is that EFSA’s latest work is likely to become the reference frame for:
In practice, if you are building EU‑facing ingestible products, you should assume that a 2 mg/day reference (or similarly conservative consumer‑use framing) will be a major anchor in 2026 compliance discussions.
The UK is moving from “managed tolerance” toward formal authorizations. The 2025 consultation signals that label conditions—especially daily intake and mandatory warnings—will become more standardized.
If you sell into Great Britain:
Consumers should always follow the label directions for the product in their jurisdiction and consult a healthcare professional for personal medical questions.
Managing cannabis compliance, licensing, regulations, and dispensary rollout is complex—and ingredient compliance is no different. If you sell ingestible CBD products across borders, you need a system that connects regulatory intelligence to label control, SKU governance, and e‑commerce logic.
Use https://cannabisregulations.ai/ to track EU and UK regulatory changes, operationalize compliant labeling by destination, and build defensible documentation for retailers, marketplaces, and enforcement inquiries.

E‑commerce has made it easy to sell the same ingestible CBD product into multiple jurisdictions—but food law does not travel as easily as parcels do. As of early 2026, operators targeting both the European Union and Great Britain face a practical contradiction in intake messaging:
For cross‑border brands, this is not just an academic debate. It affects label serving sizes, directions for use, pack architecture, online product pages, retailer onboarding, and—critically—how you defend your products in audits or enforcement actions.
This article is informational only and not legal advice.
In its updated work on CBD safety as a novel food, EFSA’s NDA Panel set a provisional safe intake level of 0.0275 mg/kg bw/day (approximately 2 mg/day for a 70 kg adult), explicitly noting that data gaps remain and that this provisional level may be revisited when better toxicology and human data become available.
EFSA’s position matters because EFSA is the EU’s risk assessor for novel foods under Regulation (EU) 2015/2283, and its scientific outputs heavily influence:
Key source:
The UK FSA (England/Wales) continues to encourage businesses to label products to reflect current consumer advice, including a 10 mg/day CBD intake for healthy adults.
The FSA’s current approach is embedded in a broader UK compliance framework that includes:
Key sources:
It is tempting to describe this as “EU stricter, UK looser.” In reality, the split reflects different regulatory moments:
The result: one consumer marketplace, two intake narratives.
Under EU novel foods rules, a product that meets the novel food definition cannot be legally marketed without authorization and inclusion on the Union list of authorized novel foods (Commission Implementing Regulation (EU) 2017/2470). The Commission’s authorization process and the Union list are here:
Even without EU‑wide authorizations, enforcement happens through national competent authorities applying:
Sources:
A key practical indicator is the EU’s Rapid Alert System for Food and Feed (RASFF), where notifications continue to flag ingestible CBD as an unauthorized novel food in specific products.
Example notifications:
If you are marketing a product as a food supplement, you still need to align with:
Practical EU supplement label expectations include a recommended daily portion and a warning not to exceed the stated portion (Directive 2002/46/EC).
Consumer-facing summary (EU):
Commission page on nutrition/health claims framework:
For England and Wales, the FSA’s Register of CBD products linked to novel food applications (often called the Public List) is the reference point many retailers use to decide what stays on shelves.
The FSA also explicitly states that enforcement decisions remain with local authorities—but the agency has recommended that products with certain statuses may stay on the market pending further consideration.
In August 2025, the FSA opened a public consultation on the authorisation of three CBD food products as novel foods, with the consultation closing 20 November 2025.
Even if you are not one of the applicants, this matters because it signals how UK risk managers may frame:
Do not overlook marketing compliance. UK regulators draw a hard line between foods/supplements and medicines. If you imply treatment or prevention of disease, you risk being treated as a medicine.
The operational problem is simple: your label’s “daily intake” language can be used against you.
If you run one pan‑regional SKU with a single set of directions, you risk:
The safest path for e‑commerce is to keep formulation constant (where possible) but tailor:
This can be executed via:
Important: within the EU, you must be cautious with geo‑targeting practices. The EU Geo‑blocking Regulation restricts unjustified discrimination, but it does not force you to sell every product into every country. You can still define delivery destinations and manage compliance risk.
Background:
At minimum, most cross‑border operators now need two intake tracks:
Operationally, this means:
The biggest hidden compliance issue is not the mg/day text—it’s that your unit dose can make compliance impossible.
If your single gummy or capsule contains 10–25 mg, you cannot credibly present an EU‑aligned serving suggestion near 2 mg/day without creating awkward fractional directions (“take 1/5 of a gummy”). That invites consumer misuse.
Common mitigation tactics:
Across both EU and UK, sophisticated retailers are increasingly asking for a “compliance dossier” that connects:
EFSA’s updated position emphasizes continuing uncertainty and the need to revisit conclusions when new data emerges. You should expect more questions about:
Reference EFSA output:
A frequent enforcement trigger is not the mg/day statement—it’s marketing copy that implies therapeutic benefit.
Key references:
Even when your focus is intake limits, enforcement often pivots to contaminants and classification.
For example, EU rules set maximum levels for certain contaminants in food, including specific contexts for hemp seed products under Commission Regulation (EU) 2023/915.
Member States may also have overlays that are stricter than EU baselines for certain product types.
EFSA has been clear that the level is provisional and dependent on future data. For operators, the actionable point is that EFSA’s latest work is likely to become the reference frame for:
In practice, if you are building EU‑facing ingestible products, you should assume that a 2 mg/day reference (or similarly conservative consumer‑use framing) will be a major anchor in 2026 compliance discussions.
The UK is moving from “managed tolerance” toward formal authorizations. The 2025 consultation signals that label conditions—especially daily intake and mandatory warnings—will become more standardized.
If you sell into Great Britain:
Consumers should always follow the label directions for the product in their jurisdiction and consult a healthcare professional for personal medical questions.
Managing cannabis compliance, licensing, regulations, and dispensary rollout is complex—and ingredient compliance is no different. If you sell ingestible CBD products across borders, you need a system that connects regulatory intelligence to label control, SKU governance, and e‑commerce logic.
Use https://cannabisregulations.ai/ to track EU and UK regulatory changes, operationalize compliant labeling by destination, and build defensible documentation for retailers, marketplaces, and enforcement inquiries.